Tainted eye drops have been linked to three deaths and eight cases of vision loss after they became infected with a drug-resistant bacteria, according to the Centers for Disease Control and Prevention (CDC).
The CDC said in a post on its website on Tuesday that the agency, along with state and local health departments, have identified 68 patients from 16 states across the country who have VIM-GES-CRPA — a rare strain of the bacteria P. aeruginosa.
The agency added that 37 cases have been linked specifically to four healthcare facility clusters.
Health officials have also gathered four reported cases of enucleation, which is the surgical removal of the eyeball, from the infection.
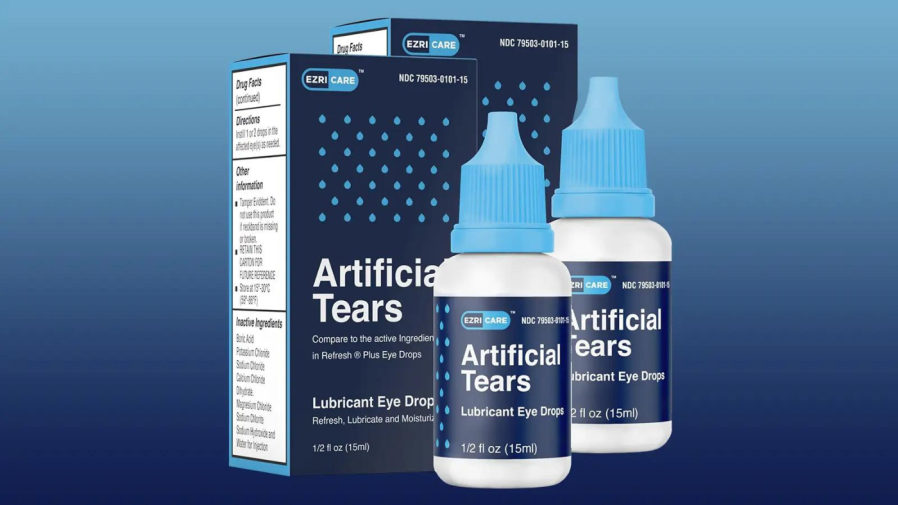
Most of the patients who have been infected said they use artificial tears, the CDC said. More than 10 different brands of artificial tears were reported as being used, but an over-the-counter product called EzriCare Artificial Tears was the most reported brand.
The CDC issued a recall for EzriCare drops and some other brands whose products have been reported to lead to the infection.
The post states that EzriCare was the only artificial tears brand that patients in each of the four healthcare facility clusters reported using. The cases were identified over a period of months, and infections were discovered in different parts of the body.
The CDC is continuing to test unopened bottles of EzriCare to determine if any contamination happened while the product was being manufactured, but the agency said patients and medical providers should not use the product at all while the testing is ongoing.
The CDC said any patients who have used EzriCare or another brand called Delsam Pharma’s Artificial Tears and have symptoms of an eye infection should seek medical care, but patients without any symptoms do not need to take any action as of the posting.
Symptoms of an infection include discharge from the eye, pain or discomfort, eye redness, feeling an object in your eye, increased sensitivity to light and blurry vision.
The Associated Press contributed to this report.